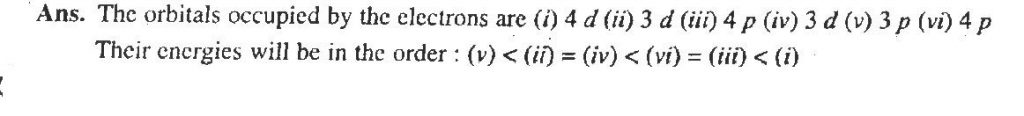The quantum numbers of six electrons are given below. Arrange them in order of increasing energies. If any of these combination(s) has/have the same energy lists: 1.n=4,l=2,mi=−2,ms=−1/2 2.n=3,l=2,ml=1,ms=+1/2 3.n=4,l=2,ml=−2,ms=−1/2 4. n=3,l=2,mi=−1,ms=+1/2 5. n=3,l=1,ml=−1,ms=+1/2 6. n=4,l=1,ml=0,ms=+1/2