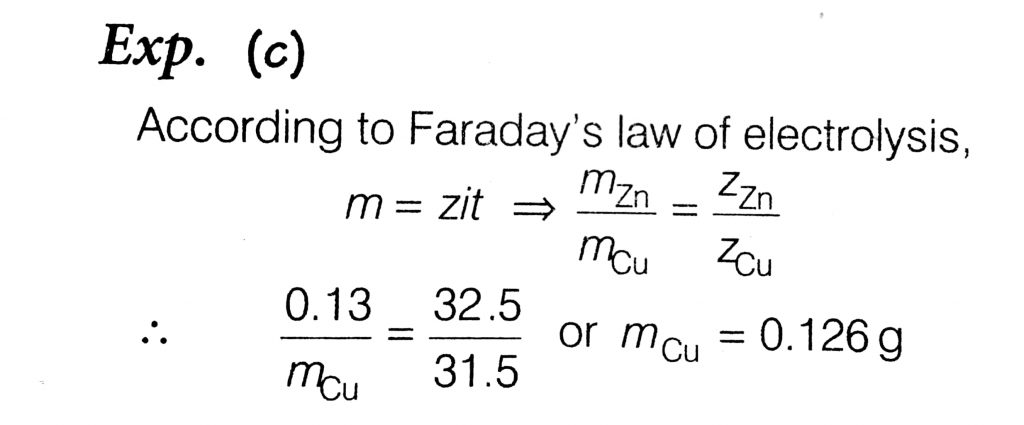The negative Zn pole of a Daniell cell, sending a constant current through a circuit, decreases in mass by 0.13 g in 30 minutes. If the electrochemical equivalent of Zn and Cu are 32.5 and 31.5 respectively, the increase in the mass of the positive. Cu pole in this time is