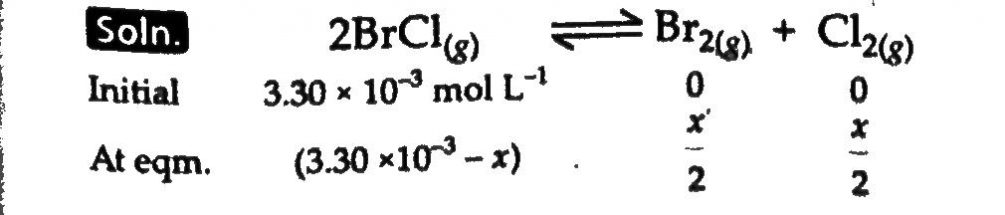Bromine monochloride, BrCl decomposes into bromine and chlorine and reaches the equilibrium 2BrCl(g)→Br2(g)+Cl2(g) For which Kc = 32 at 500K. If initially pure BrCl is present at a concentration of 3.3×10^−3 molL^−1 , what is it’s molar concentration in the mixture of equilibrium?